Nanopatterned glass could yield medical implants with lower rejection risk

Experiments by scientists at Yale University suggest that arrays of ultra tiny rods made of complex alloys called bulk metallic glasses (BMGs) could be used to produce medical implants with lower risk of rejection, the researchers report.
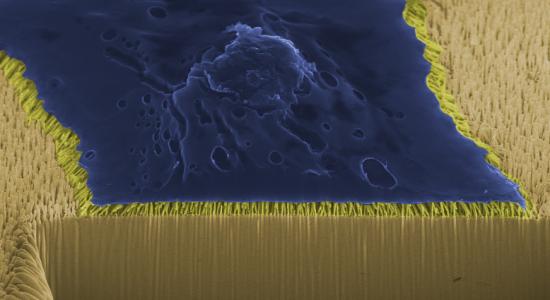
BMGs are a category of strong but extremely pliable glass. The scientists showed that they could manipulate human immune cell response to foreign substances — such as the material in a heart stent — by covering the surface with the tiny BMG rods. The tiny rods are called nanorods.
“BMGs and their unique processability allow us to investigate cell interactions at an unprecedented nanoscale resolution,” said Themis Kyriakides, associate professor of pathology and biomedical engineering and principal investigator of the research, published in the journal ACS Nano.
The team’s array of BMG nanorods, formed on penny-sized BMG discs, varied in diameter from 55 nanometers to 200 nanometers — nearly a thousand times narrower than a human hair and narrower than can be made using other materials.
The researchers focused on how differing nanorod arrays affected three cell types: macrophages, fibroblasts, and endothelial cells, all of which help recognize and respond to foreign materials in the human body. After growing each cell on a BMG disc for 24 hours, the researchers observed each cell’s reaction to the disc’s specific array, or “nanopattern.”
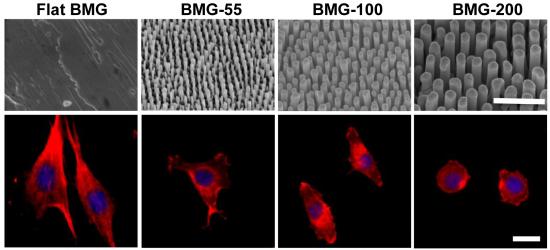
The fibroblasts, for example (pictured), decreased in size and became more circular when grown on nanopatterned BMGs. “This finding is key because cell spreading and shape are correlated to function,” said first author Jagannath Padmanabhan, a doctoral student in biomedical engineering. “And in fact the fibroblast cells, which normally break down a material by encapsulating it in collagen, produced significantly less collagen when grown on nanopatterned substrates. Such results suggest that nanopatterned biomaterials could help prevent implant failure.”
However, the researchers noted that each cell type responded differently to the nanopatterns, and some cells didn’t respond at all to the smallest nanorod arrays. “The eventual hope,” said Padmanabhan, “is that you could design one pattern that simultaneously engineers multiple cell types to do different things.”
That would be good news for patients, said Jan Schroers, professor of mechanical engineering & materials science and senior co-author of the research. “Using BMGs as a material, we can simultaneously create the intricate structures of nanorods and the larger, complex shapes of heart stents and medical sensors,” he said. “Combining nanopatterns with the fabrication of medical devices could lead to implants with lower rejection rates than even current ‘state-of-the-art’ technologies.”

Other Yale authors include mechanical engineering doctoral student Emily Kinser, research intern Christopher Duncan-Lewis, postdoctoral fellow in anesthesiology Jenna Balestrini, and postdoctoral fellow in pathology Andrew Sawyer; Mark Stalter of the IBM Semiconductor Research and Development Center also contributed to the research, which was published on April 11.
“We are grateful,” said Kyriakides, “to be part of the Yale Center for Research on Interface Structures and Phenomena, which encourages such bridges between nanotechnology and medicine.”

