A Better Way To Study Batteries, From The Transformative Materials & Devices Lab
If lithium-air batteries live up to their promise, we could one day be driving electric cars 500 miles or more without recharging, or using laptops for weeks without having to plug in. They could also replace lithium-ion batteries, currently the standard in many consumer electronics.
 But their chemistry has proven frustratingly hard to understand, and researchers need to learn more before the batteries can break out of the lab and onto the market. Researchers from Yale and MIT believe that a laser analysis technique known as Raman spectroscopy could answer many of the questions that remain about the batteries, also known as lithium-oxygen or Li-O2. Their paper was published online July 31 by the journal ChemElectroChem (it will be published in the print edition in October). The paper was designated a “Very Important Paper” by the journal's editor.
But their chemistry has proven frustratingly hard to understand, and researchers need to learn more before the batteries can break out of the lab and onto the market. Researchers from Yale and MIT believe that a laser analysis technique known as Raman spectroscopy could answer many of the questions that remain about the batteries, also known as lithium-oxygen or Li-O2. Their paper was published online July 31 by the journal ChemElectroChem (it will be published in the print edition in October). The paper was designated a “Very Important Paper” by the journal's editor.
“Lithium-oxygen is quite superior to lithium-ion batteries, at least in theory,” said lead author Forrest S. Gittleson, a post-doctoral associate at Yale in Chemical & Environmental Engineering. “It can store at least two to three times the energy that you can store in lithium-ion.”
For a battery to be rechargeable, though, the changes that convert chemical energy to electrical energy must be efficiently reversible. Among other obstacles, lithium-oxygen systems have not yet demonstrated high-energy reversible cycling, so recharging is a problem. Before they can solve this, researchers need to learn more about its mechanisms.
“This is an extremely new technology, and not all of its chemistry is very well understood,” Gittleson said.
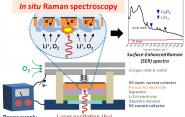
Partly because it allows for real time (or in situ) analysis, the researchers say that Raman spectroscopy might be the best tool to do this. Unlike a handful of commonly used spectroscopic and diffraction techniques, Raman spectroscopy can provide a comprehensive look at the electrochemical processes in the systems of lithium-air batteries. In a study published last year, the authors observed the production of reversible lithium superoxide, but also some side products that could hinder charging. The in situ techniques of Raman spectroscopy allow researchers to directly observe the formation and decomposition pathways, and gain insight into lithium-oxygen chemical mechanisms.
“It’s certainly promising for advancing the science,” Gittleson said. “And it’s particularly promising that you can discern the specific species with confidence.”
In use since 1928 (although not with lasers until 1960), Raman spectroscopy uses light to detect molecular vibrations in polarizable species. When a laser pulse is applied to a sample, molecules are excited to virtual energy states. These molecules then relax and emit photons, which can be detected. The behavior of those photons allows for a “molecular fingerprint.”
It’s a fairly common tool, used by everyone from art conservators to authenticate artwork to law enforcement officials to detect homemade drugs. But it’s been slow to catch on in the battery research world, Gittleson said.
“Not every lab has access to Ramen spectroscopy equipment, and not everyone working on batteries realizes the potential yet,” he said.
André D. Taylor, director of the Transformative Materials & Devices Lab at Yale and senior author of the paper, said he hopes that this will change.
“This is a powerful technology, and we want to get people to see this as a viable technology,” he said.
Gittleson said there are still a lot of challenges before lithium-air batteries can be put to practical use – the instability of its electrolytes among them. But with more widespread use of Raman spectroscopy, researchers will have a much better understanding of its internal chemistry – and that’s critical to advancing the development of the technology.
The paper’s other authors are Won-Hee Ryu, a post-doc in Taylor’s lab, and from MIT, Koffi P. C. Yao, David G. Kwabi, Sayed Youssef Sayed and Yang Shao-Horn.

