IRP Focus: Surfaces & Nanomaterials: Research Projects
Striving for a Solar Cell Efficiency Record

Minjoo Larry Lee, Dept. of Electrical Eng.
Powering homes with clean, sustainable solar energy has long been an appealing and popular idea, but the shortcomings of current technology still stand in the way of the widespread use of solar cells. Most commercially available solar cells today convert solar energy to electricity at efficiency levels of only between 10 and 29 percent.
Developing New Catalysts from Ferroelectric Materials
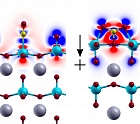
Eric Altman, Dept. of ChE & EnvE
The role of a heterogeneous catalyst, used in essentially every industrial chemical process, is to speed up a chemical reaction by providing a surface where that reaction readily occurs. By investigating novel surfaces, understanding their properties, and transforming them at the atomic level, the Altman Lab is making headway on developing entirely new categories of catalysts with potential to impact a variety of industries.
Using the Force of Light to Power Nanomachines

Hong Tang, Dept. of Electrical Eng.
In recent years, members of the Tang Lab have won acclaim for describing the forces of light that can both attract and repel. This force is very weak, but its pressure is powerful enough to push an object of nanometer size and picogram mass, making it a potentially ideal force for controlling nanodevices.
Engineering Speedier Silicon Chips with Quantum Dots

Minjoo Larry Lee, Dept. of Electrical Eng.
Imagine holding a crystal in the palm of your hand, perhaps an exotic specimen from the Natural History museum or something as mundane as rock candy. Then you cut it down to half its size over and over again. By the time you’ve halved its size about 20 times, you are left with nanoscale particles so tiny as to be considered "zero dimensional.” When the nanoscale bits are composed of semiconductor materials, they are known as quantum dots, and their properties can diverge widely from larger samples of the same material. One of the most remarkable aspects of quantum dots is their ability to trap or “confine” charge carriers and to convert them into light.
Developing Efficient, Commercially Viable Fuel Cells
André Taylor, Dept. of ChE & EnvE; Jan Schroers, Dept. of Mech. Eng, & Mats. Sci.
Fuel cells have been touted as a cleaner solution to tomorrow's energy needs, with potential applications in everything from small portable electronic devices to automobiles to industrial facilities. But one reason fuel cells aren't already more widespread is their lack of endurance. Over time, the catalysts used even in today's state-of-the-art cells break down, inhibiting the chemical reaction that converts fuel into electricity. In addition, only a fraction of the catalyst is properly utilized at any given time, while much of it sits tucked away, unexposed and unavailable for chemical reaction.
Investigating Atomic Precision at the Oxide Interface
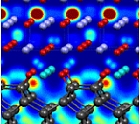
Eric I. Altman, Dept. of ChE & EnvE; Sohrab Ismail-Beigi, Dept. of Applied Physics, Dept. of Physics
For many materials scientists, what goes on at the interface between two different materials is where the excitement is. Long gone are the days when electronics developers glued materials together by hand. Today’s materials are sandwiched together with atomic-scale precision – an advancement that has led to the control of exotic solid-state phenomena, such as magnetism and superconductivity at the nanoscale and a promise of applications that will have broad-sweeping impact on the technologies of our time.
Exploiting the Novel Properties of Bulk Metallic Glass

Themis Kyriakides, Dept. of Biomedical Eng., Yale School of Medicine; Jan Schroers, Dept. of Mech. Eng. & Mats. Sci.
In the field of biomedical implants, device developers rely on materials whose surface properties can be controlled and modified, that can function in nanoscale devices, and that are biocompatible. Having struck on novel methods for exploiting the unique properties of metal alloys known as bulk metallic glasses, mechanical engineers and materials scientists in the Schroers Lab are collaborating with biomedical engineering colleagues in the Kyriakides Lab to expand that materials’ application to medical uses.
Developing New Materials at Warp Speed

Jan Schroers, Dept. of Mech. Eng. & Mats. Sci.
In recent decades, high-throughput screening of chemical compounds revolutionized the pharmaceutical industry. Now, Yale engineers are set to revolutionize the understanding and development of materials with a technique they’ve dubbed “high-throughput characterization.”
Perfecting and Simplifying the Fabrication of a New Semiconductor

Jung Han, Dept. of Electrical Eng.
For more than a decade, gallium nitride has been a focus of semiconductor research and a challenging target for materials scientists who seek to exploit its unique properties. The superior energy efficiency of GaN-light-emitted diodes is already displacing conventional lighting technologies, and GaN transistors are predicted to have ubiquitous applications in modernizing our power-grid and electricity infrastructure.
Addressing Key Challenges in Biomaterials
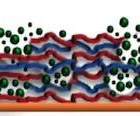
Paul Van Tassel, Dept. of ChE & EnvE
Biomaterials are ubiquitous in modern medicine, from prosthetic limbs to device coatings, and hold promise for future applications like scaffolding for tissue regeneration. Biomaterial design presents a key challenge, however: applications often require materials to be both mechanically rigid (to promote strong cell adhesion) and bioactive (i.e. able to communicate specific cues to contacting cells). To date, achieving one of these features usually required sacrificing the other.

