IRP Focus: Energy & Sustainability: Research Projects
Perfecting and Simplifying the Fabrication of a New Semiconductor

Jung Han, Dept. of Electrical Eng.
For more than a decade, gallium nitride has been a focus of semiconductor research and a challenging target for materials scientists who seek to exploit its unique properties. The superior energy efficiency of GaN-light-emitted diodes is already displacing conventional lighting technologies, and GaN transistors are predicted to have ubiquitous applications in modernizing our power-grid and electricity infrastructure.
Developing New Catalysts from Ferroelectric Materials
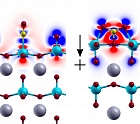
Eric Altman, Dept. of ChE & EnvE
The role of a heterogeneous catalyst, used in essentially every industrial chemical process, is to speed up a chemical reaction by providing a surface where that reaction readily occurs. By investigating novel surfaces, understanding their properties, and transforming them at the atomic level, the Altman Lab is making headway on developing entirely new categories of catalysts with potential to impact a variety of industries.
Unraveling Microalgae Genetics to Perfect Biofuels

Jordan Peccia, Dept. of ChE & EnvE
Though the idea of producing biofuels from algae is not new, scientists as yet know little about the genes that algae use to make lipids—fats that can be used as biofuel—or how they can be turned off and on under different environmental conditions. As attention to alternative energy sources increases, however, renewed attention is being paid to the genetic makeup of microalgae.
Forward Osmosis for Clean Water and Energy

Menachem Elimelech, Dept. of ChE & EnvE
Water and energy are two resources on which all of modern society depends. As demands for each increase, researchers look to alternative technologies that promise sustainability and reduced environmental impact.
Developing Efficient, Commercially Viable Fuel Cells
André Taylor, Dept. of ChE & EnvE; Jan Schroers, Dept. of Mech. Eng, & Mats. Sci.
Fuel cells have been touted as a cleaner solution to tomorrow's energy needs, with potential applications in everything from small portable electronic devices to automobiles to industrial facilities. But one reason fuel cells aren't already more widespread is their lack of endurance. Over time, the catalysts used even in today's state-of-the-art cells break down, inhibiting the chemical reaction that converts fuel into electricity. In addition, only a fraction of the catalyst is properly utilized at any given time, while much of it sits tucked away, unexposed and unavailable for chemical reaction.
Striving for a Solar Cell Efficiency Record

Minjoo Larry Lee, Dept. of Electrical Eng.
Powering homes with clean, sustainable solar energy has long been an appealing and popular idea, but the shortcomings of current technology still stand in the way of the widespread use of solar cells. Most commercially available solar cells today convert solar energy to electricity at efficiency levels of only between 10 and 29 percent.
Designing Functional Proteins that Move Electrons
Corey J. Wilson, Dept. of ChE & EnvE
Caltech theoretical chemist Rudolph A. Marcus won the Nobel Prize in 1992 for his contributions to the theory of electron transfer reactions in chemical systems. But he did not present an instruction manual for replicating them.

